The Mid-America Organic Association Conference and Expo was held here in Kansas City last week. Farmers, gardeners, agriculture experts, and consumers came from all over the mid-west to share their commitment to clean, healthy, chemical-free, naturally grown food crops. Manufacturers showcased many innovative and environmentally sound products and services. The exchange of information and ideas was incredibly inspiring and created an atmosphere that was nothing short of electric.
Each individual we spoke with had a passion for cooperating with Nature in the growing of our food and was firmly committed to healing and caring for the land. We met with speakers such as David Yarrow, sustainable farming researcher and advocate, and Joel Adams of Adams’ Osage Ridge Aronia Community Farm, who are both tireless in their endeavors to farm, research, educate, innovate, and bring together many schools of agricultural thought for the purpose of growing the highest quality food possible.
We were privileged to spend an hour with Jim Porterfield, a brilliant agricultural researcher who has been involved in cutting edge science and innovation in growing nutrient dense crops. We visited a number of vendor booths that featured products with an emphasis on trace minerals combined with humates and inoculants. We witnessed the “mineral message” gaining momentum among organic farmers and those who supply them with carbon building inputs.
We also met with farmers like Michelle from Illinois, mother and homeschooler of five, who was excited to learn about soil amendments and how they could help her transform an acre of clay-heavy soil into a minerally balanced, sustainable and diverse teaching garden for her children. And we met with Tasha from St. Louis, a community activist who is passionate about turning vacant urban lots with poor soil into community gardens. Frank LeBeau, Garden Educator with Heal the Planet Farm, stopped by to chat about the need for mineralizing the soil at their 350-acre organic permaculture farm located at the base of the Ozark Mountains in Southern Missouri, an area where traditional farm acreage suffers from depleted soil.
This diverse group of individuals and companies made it profoundly clear that healthy, nutrient-dense, sustainably grown, humanely raised food is imperative to the well being of our families and communities, our country and our planet. But paramount to all this, is the sobering truth that this can only be accomplished by us. Yes, US!
It will take consumers directing the resources of their pocket books toward the purchasing of this food. Our organic and biodynamic farmers stand enthusiastically ready to produce more and more for the growing demand. Existing organic markets will thrive and new ones will be established. Supermarkets will become dominated by ever-larger “organic departments,” until one day we are daily nourished by the quality of healthy food once enjoyed only by our ancestors.
These necessary changes in the way we produce our food cannot be left up to our government or multi-national agribusiness corporations. WE must direct their policies and actions. That which the consumer demands ultimately determines what is produced and how it is produced.
Mary had completed that all-important first step—taking a soil sample. The laboratory analysis had been received, the custom soil Rx had been written, and the soil amendment blend was created for her specific soil needs.
It was show time in the Wenger garden on December 4th as the soil amendment blend was broadcast over the loose soil and tilled in to the top six inches. That top six inches of soil, often referred to as the plow layer, is where most of the root growth and nutrient exchange takes place.
December 4th might seem to be a late date, but our mid-western weather was very mild this autumn, and Mary was pulling late season greens and potatoes from her garden well into November. The ground had not yet frozen, and she wanted to take advantage of winter snow and rain to help incorporate the new amendments.
Mary had actually delayed the operation by a day due to excessive wind, which can blow away many of the fines before they get worked into the soil. She watered the amendments in and covered the soil with a light layer of hay.
Bringing your soil into optimum balance does often require an investment. Mary utilized a wise option in soil balancing, which is to treat half of your garden, field, or pasture one year and the other half the next year. There are two main advantages to this. First, it spreads the cost over two years and second, it gives you an untreated (control) area that illustrates how much better the treated area produces in terms of quality, nutrient-density, and yield. Depending on the size of your project, you may decide to balance the soil incrementally over several growing seasons.
This time of year is a great time to peruse the seed catalogs that are coming out and laying some plans for the upcoming growing season. With that in mind, we discussed the benefits of keeping the soil covered at all times and how that sequesters carbon, reduces erosion, and provides free, amino acid Nitrogen for next growing season. Plans are being made for late winter/early spring cold hardy cover crops to be planted, providing that Nitrogen source as well as a “plant through mulch” after cutting down the cover crop before it goes to seed.
We will keep you updated with the progress of Mary’s garden. Till next time … #soilminerals #soilamendments #covercrops www.sustainablesoils.com
Last May, we had the pleasure of meeting a new customer, Mary Wenger, who along with her family raises her own produce on a 1,750 square ft. backyard garden.
We have often written about minerals and their importance to health, vigor, nutrient-density, and yield, and it was a pleasure to share this message with Mary in person.
With this in mind, we thought we would follow the progress of Mary’s garden to illustrate how mineral balance works, and what the results are. Beginning with an examination of the garden’s current quality and productivity, we will chart its progress and follow the improvements in crop nutrient-density and mineral content of plant sap through the use of a refractometer. We will also illustrate how a mineral balanced, biologically active soil greatly reduces and even eliminates weed, pest, and disease problems.
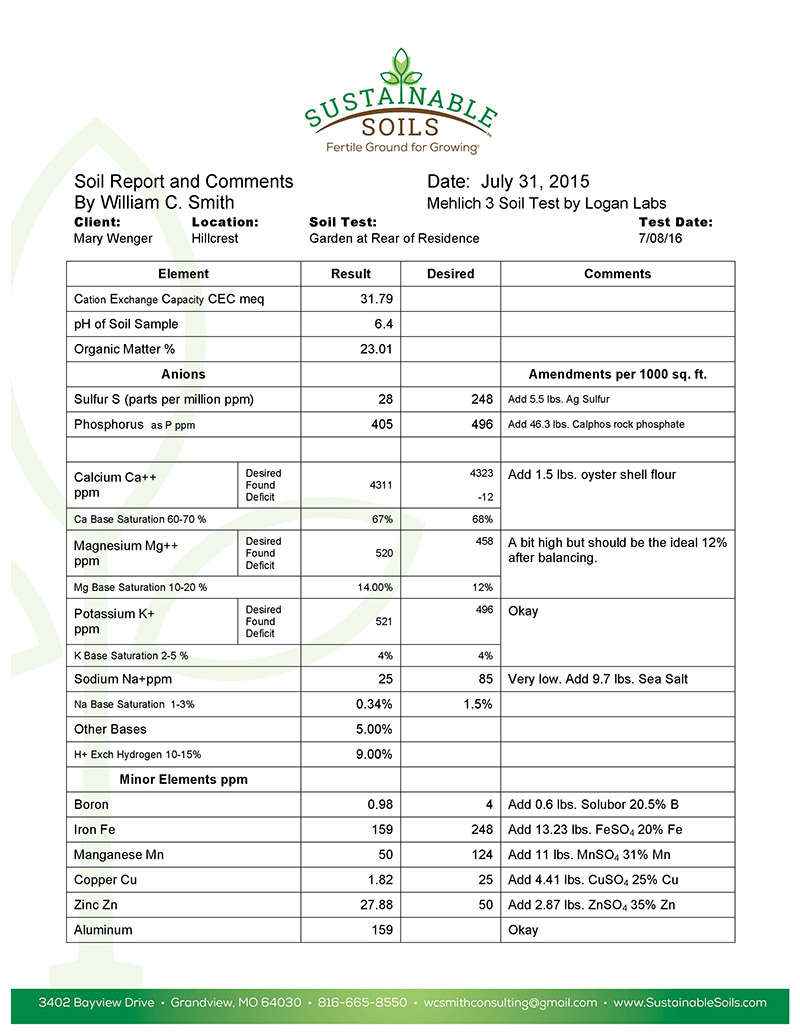
The first step, and it’s always the first step, is to get a standard soil test. You must know where you are to get where you want and need to go. As we move through the process of balancing Mary’s soil, we’ll post interviews with Mary and her family as we monitor the progress.
Let’s begin this journey by looking at the soil test results and the soil Rx showing what we need to bring this soil into optimum balance. Next time, we will examine in detail the soil amendments we use and what we accomplish with them. If you would like to see instructions on how to take a soil sample, and view more soil prescriptions, you can visit our website at: http://sustainablesoils.com/get-started/ and http://sustainablesoils.com/wp-content/uploads/2015/10/SampleSoilPrescriptions.pdf

Although the metal Manganese is gray in color in its pure form, the Incas called the reddish gemstone, rhodochrosite, Inca Rose, believing that this Manganese and carbonate-based substance was the blood of their former kings and queens turned to stone. Perhaps they had a clue as to how essential Manganese is to all living organisms.
Arden Anderson, doctor, farmer, and author of “Science in Agriculture,” long ago made the connection between nutrient-dense foods grown in mineral rich soils and the health benefits they offer us. He suggests Manganese provides the electrical charge that draws other elements into the seed. There is an atom of Manganese at the center of the germ of every seed and Manganese is necessary for reproduction through the formation of healthy seeds.
Manganese plays a very crucial role in plants and is necessary in nitrogen metabolism, photosynthesis, and the formation of other compounds required for plant metabolism. It also acts as an activator for more than 35 different enzymes. Manganese is a synergist with iron. They work together and need each other. An example of this interdependence is that high levels of available iron in the soil can limit the uptake of Manganese.
Manganese, proposed to be an element by Carl Wilhelm Scheele in 1774, was actually discovered by the Swedish chemist Johan Gottlieb Gahn when he heated the mineral pyrolusite (MnO2) in the presence of charcoal. Today, Manganese for agricultural use is obtained from pyrolusite by treating it with sulfuric acid (H2SO4) to form manganese sulfate (MnSO4), which is then electrolyzed.
Manganese is essential to all plants. However, certain crops have been found to be especially responsive to adequate levels of Manganese such as beets, cauliflower, citrus, lettuce, onions, potatoes, spinach, sweet corn, small grains, large-seeded legumes, alfalfa, sorghum, soybeans, cotton, and tobacco.
Manganese is not translocated in the plant so deficiency symptoms appear first on younger leaves. Interveinal chlorosis is the most common symptom and in severe cases, a series of brownish-black specks appear in the affected areas with eventual premature leaf drop. Delayed maturity is another deficiency symptom in some species. Manganese deficiency is prevalent in calcareous soils, high pH soils, and soils with excessive levels of organic matter. Excessive levels of Calcium and Magnesium can also interfere with Manganese uptake.
Manganese toxicity can become a problem in soils deficient in Copper and Zinc. The late John Mark Purdey, an organic farmer and brilliant researcher, found that bovine spongiform encephalopathy (BSE) or mad cow disease and chronic wasting disease, are directly correlated with soils very low in Copper and Zinc in combination with high levels of Manganese. In this worldwide study, he discovered that BSE and CWD may not be the result of viral infections but the result of a mineral imbalance.
Both animals and humans require Manganese. In humans, Manganese activates the enzymes necessary for the body to use biotin, thiamine (B1), vitamin C, and choline. It’s also needed for healthy nerves, immune system function, blood sugar regulation, formation of mother’s milk, and the growth of healthy bones. Deficiency may lead to trembling hands, seizures, and lack of coordination. Manganese is a co-factor for an enzyme called manganese superoxide dismutase (MnSOD), an antioxidant associated with protection against free radical damage. Conditions marked by increased free radical damage to cells and tissue have been linked to diets low in manganese. In animal nutrition, an excess of manganese increases the need for iron, and a manganese deficiency results in leg deformities in calves, eggs not formed correctly, degeneration of testicles, offspring born dead, and delayed heat periods. An excess of calcium and phosphorus may lead to a manganese deficiency.
It is quite clear that animals and human beings need adequate levels of Manganese. For this to occur, it must be in the soil in adequate levels and in optimum balance with Sulfur, Phosphorus, Calcium, Magnesium, Potassium, Sodium, Boron, Iron, copper, and Zinc. For all of this to work and result in maximum animal and human nutrition, trace elements such as Chromium, Cobalt, Iodine, molybdenum, Selenium, Tin, Vanadium, Nickel, and Fluorine must also be present in tiny amounts. Deficiencies in these trace minerals can also be problematic but that’s a different story for another time.
Sources:
Astera, Michael. With Agricola. The Ideal Soil 2014: A Handbook for the New Agriculture v2.0. Michael Astera, 2014. Print.
Walters, Charles, and C. J. Fenzau. Eco-Farm. Acres USA, 1996. Print.
Andersen, Arden. Science in Agriculture. Acres USA, 2000. Print.
DVM. Skow, Dan, and Charles Walters. Mainline Farming for Century 21. Acres USA, 1995. Print.
Fallon, Sally, with Mary Enig. Nourishing Traditions. New Trends, 2001. Print.
Phosphorus was the 13th element to be discovered, and the first that was not known since ancient times. For this reason, and also due to its use in explosives, poisons and nerve agents, it is sometimes referred to as “the Devil’s element.”1, 2 German alchemist Hennig Brand is credited for discovering this element in 1669 when he distilled approximately 5,500 liters of human urine to produce 120 grams of a glowing substance which he ultimately named Phosphorus, from the Greek word for “light-bearing.”
For over a century, Phosphorus has had multiple functions in soil chemistry. Today we know that optimum levels of Phosphorus enhance early root formation and growth, flowering and seeding, uniform crop maturity, and stalk and stem strength. It is important for photosynthesis and respiration. Respiration is the process by which plants convert photosynthates such as sugars, back into energy for growth and other metabolic processes.
Soil Phosphorus is immobile for the most part and the level of soluble Phosphorus immediately available to plants is rather small. In order for plants to satisfy their need for Phosphorus, their root systems must explore large areas of soil. Mycorrhizal fungi greatly assist plant root systems in this exploration by forming a symbiotic relationship with them. The thread-like hyphae of the fungus connect with plant root hairs and extend great distances into the soil, taking up nutrients and transporting them back to the plants. This results in increased Phosphorus uptake. In exchange, the mycorrhizal fungi receive sugars and other compounds from the roots.
The uptake of Phosphorus can be limited by soil compaction, pH, and moisture. Even in high Phosphorus soils, anything that inhibits aggressive root growth, such as compacted soil, will have an adverse effect on Phosphorus uptake. With a pH greater than 7.0, Phosphorus can be fixed in less available forms by excess Calcium. With a pH of less than 6.0, Phosphorus can be fixed in less soluble forms by excess soluble Aluminum. Phosphorus availability is at its highest at pH 6.4 to 6.8. Optimum levels of Phosphorus will result in higher Phosphorus uptake at all moisture levels. However, moisture stress results in lower Phosphorus availability. Moisture levels exceeding soil capacity exclude the oxygen required for Phosphorus uptake.
Phosphorus is a highly reactive element and does not exist in elemental form in soil. Plant roots take up nearly all Phosphorus as phosphate. (P2O5) Orthophosphate is the simplest phosphate with the chemical formula PO4-3. Primary orthophosphate has a chemical formula of HP204–, is dominant in acidic soils, and is taken up much more readily than the secondary form, HPO42- that is the dominant available form of Phosphorus in alkaline soil conditions. All Phosphorus sources added to soil must be converted to the orthophosphate forms before plants can utilize them.
Organic, biodynamic, and biological farming/gardening practices utilize a direct application of rock phosphate as the source of Phosphorus. Colloidal soft phosphate rock and Tennessee Brown Phosphate rock are inexpensive, commonly available Phosphorus sources, both of which are approved for organic use by the USDA National Organic Program. Phosphate rock is a largely insoluble, slow release Phosphorus source. Only about 3% orthophosphate is soluble and readily available to plants. The rest remains “locked up” or chemically bonded to Calcium and is gradually released for plant use by the action of soil microbes. So the Phosphorus is long lasting, remains insoluble and in reserve, and is not subject to leaching. Optimum levels of phosphate rock in the soil contain enough “reserves” to provide the release of available Phosphorus in more than adequate quantities for early season requirements, as well as a continuous supply throughout the growing season.
Phosphate rock is also the raw material used to make highly mobile, water soluble Phosphorus fertilizers (WSP fertilizers) used by large scale farming operations. The global fertilizer industry consumes nearly 90% of the world’s phosphate rock production in the manufacture of WSP fertilizers. The remainder is used to make elemental phosphorus and animal feed supplements, or is applied directly to soils. Phosphate rock and sulfuric acid are the raw materials used in the manufacture of single superphosphate (SSP) and phosphoric acid. Phosphoric acid is an intermediate by-product used to make triple superphosphate (TSP) and ammonium phosphate. Highly concentrated, compound NPK formulations now form the mainstay of the global fertilizer industry (Engelstad and Hellums, 1993; UNIDO and IFDC, 1998).
Unfortunately, the high solubility of these concentrated NPK fertilizers, and their overuse in efforts to achieve huge yields of big, tall, expanded but nutrient deficient crops, is very detrimental to our environment. Unlike phosphate rock, which stays in the soil and releases its “reserves” of Phosphorus over time, superphosphate fertilizer is subject to leaching and runoff. Extensive use of concentrated NPK fertilizers destroys microbes, earthworms, and other beneficial soil organisms. The soil loses its beneficial texture and becomes less capable of absorbing water. Topsoil erosion becomes problematic and the runoff of rain carries soil particles, and the Phosphorus attached to them, into streams, rivers, and lakes.
Unnaturally high Phosphorus levels in these bodies of water fuel excessive growth of algae. In a process called “eutrophication,” these algae die, and are decomposed by bacteria that use dissolved oxygen. This creates “dead zones” resulting in fish kills, surface water scum, decreased recreational use, and foul odors.
Most phosphate rock is mined using large-scale surface methods. Michael Astera of Agricola has written one of the most insightful concepts regarding the mining of all minerals used in agriculture. It appears as follows:
“Mining of the needed minerals need not entail long-term environmental damage
either. Mines and quarries can be carefully worked by those who care about their
home planet, and when the mines are depleted they can be landscaped and
planted to be as or more beautiful than before mining. It’s also worth noting that
many of the economically viable sources for agricultural minerals contain such
high concentrations of these minerals that they are toxic to soil life and little or
nothing grows there. Removing these toxic concentrations and using them to
make other parts of the planet healthier and more productive can, at the same
time, open up these formerly toxic soils to the growth of forest or grasslands.
None of this should be done on the basis of greed or short-term gain, but rather
wisely, intelligently, and in harmony with Nature.”
Phosphorous, when present along with 10 other important minerals, all in ideal ratios to each other, results in a fertile soil capable of producing vigorous, nutrient-dense crops of superior quality. Such a mineral balance also works to sequester carbon, improve soil texture and drainage, provide adequate water retention, prevent erosion by water or wind, promote biological activity, and reduce costly annual inputs. Balancing the minerals in the soil is the first step toward sustainability because if the minerals are not in the soil, they can’t be in the food or recycled on the farm or in the garden.
Organic, biological, and biodynamic farming and gardening systems strive to reduce “outside inputs.” Once the minerals in the soil are balanced, you won’t have to add them in those quantities again. A soil test in the spring and fall will indicate any minor adjustments needed. Sulfur and Boron will be the minerals most likely needing attention as plants readily take them up. The superior health of a minerally balanced soil greatly reduces the need for inputs other than these minor adjustments. Since the minerals are in the soil, they will be in the crop residues, cover crops, and livestock manure and will be recycled through these sustainable systems.
Justus von Liebig, considered to be the founder of organic chemistry, popularized The Law of the Minimum. “The availability of the most abundant nutrient in the soil is only as good as the availability of the least abundant nutrient in the soil.” Whichever nutrient is in the shortest supply will limit the performance of the crop. This illustrates why balance, and optimum quantities of mineral nutrients, is so vital.
The Farming Systems Trial (FST) at Rodale Institute, is America’s longest running side-by-side comparison of organic and conventional agriculture. Over thirty five years of scientific data from the FST have proven that organic systems produce yields equal to conventional, outperform conventional yields in years of drought, build rather than deplete soil, and are actually more profitable than conventional farming.
We have focused on Phosphorus and its importance to sustainability. We have discovered that it takes a synergy of Phosphorus and ten other mineral nutrients to maximize soil fertility, and we have looked at just a few of Rodale Institute’s FST empirical, scientific findings. Moving forward in sustainability calls for the coming together of many ideas, scientific facts, and schools of thought.
1 Emsley, John (7 January 2002). The 13th Element: The Sordid Tale of Murder, Fire, and Phosphorus. John Wiley & Sons. ISBN 978-0-471-44149-6. Retrieved 2016-08-26.
2 Weeks, Mary Elvira (1932). “The discovery of the elements. II. Elements known to the alchemists”. Journal of Chemical Education. 9: 11. Bibcode:1932JChEd…9…11W. doi:10.1021/ed009p11.
“We know more about the movement of celestial bodies than about the soil underfoot.”
– Leonardo DaVinci
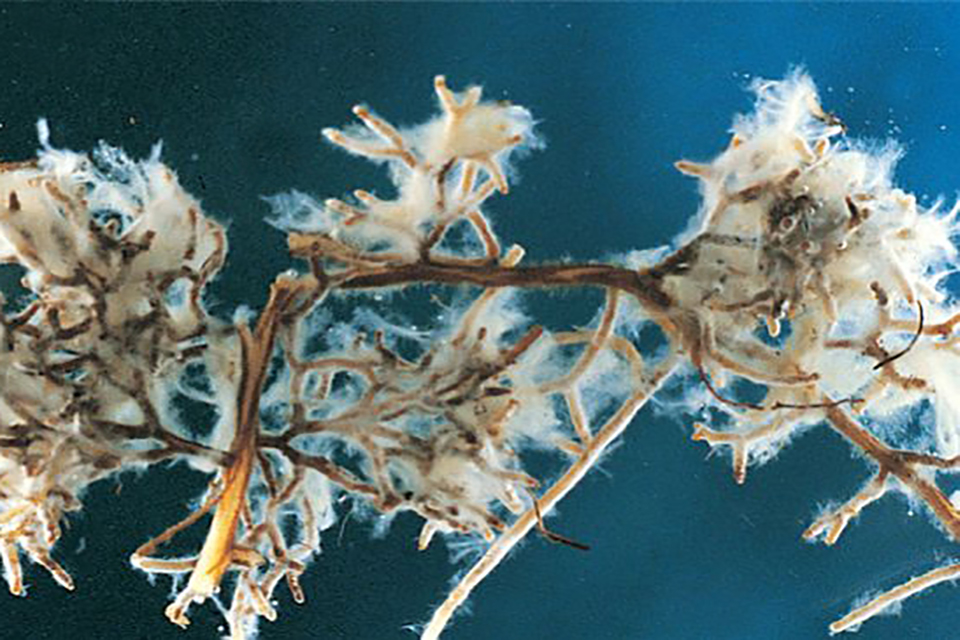
Biological activity in the soil is imperative to the development of vigorous, disease- and pest-resistant crops in the garden or field. Fungi, and bacteria are beneficial soil organisms that have a symbiotic relationship with the plant through its root system. They also play a major part in the breakdown of organic matter and the freeing of nutrients that may be in the soil, yet unavailable to plant roots. Some fungi even produce antibiotics that protect the plant from disease.
95% of plant species have a relationship with fungi. Mycorrhizal (my-ko-rize’-ul) fungi have an elaborate root system of fungal filaments that grow yards, both deep into the soil and in the root zone. Several miles of these filaments can be found in a few grams of soil. Mycorrhizal fungi seek out moisture and nutrients and bring them back to the plant roots. Therefore, the surface absorbing area of plant roots can be increased hundreds of times.
Myco means fungus. Rhizae comes from rhizome, meaning roots. This translates into fungus root. There are two types of mycorrhizae, (my-ko-rize’-ee) Endo-mycorrhizae, and ecto-mycorrhizae. Endo-mycorrhizae (meaning inside) actually penetrate the tiny root hairs of plants without changing the root’s cell structure, effectively extending the plants root system. The fungi bring moisture and nutrients back to the roots and in turn, the roots share sugars and other carbohydrates with the fungi. The fungi also release powerful enzymes that dissolve, and make available, nutrients that are tightly bound and difficult to access. Phosphorous, for example, a very active mineral with a strong negative charge can bond with Calcium, which has a double positive charge. The result is Calcium phosphate, a very stable compound. The enzymes break the Calcium/Phosphorous bond making the Phosphorous available for uptake by the plant roots. 90% of cultivated plants have a symbiotic relationship with endo-mycorrhizae.
Ecto-mycorrhizae fungi (meaning outside) grow next to plant roots but live completely outside of the roots. They share nutrients with the plant roots through the soil water/nutrient solution.
Certain types of bacteria, called rhizobia, live in symbiosis with legumes such as red clover, cowpeas, and alfalfa. These bacteria form and inhabit nodules on the roots of legumes and are able to take Nitrogen from air in the soil and create ammonia, which the plant uses to create amino acids and other compounds. The rhizobia in turn receive carbohydrates from the plant. These Nitrogen-gathering or Nitrogen-fixing bacteria are not found on common crop plants except legumes. Green manure is a rich source of Nitrogen for the soil and comes from turning these legumes under in your garden or field.
Some soil inoculant products contain numerous species of endo-mycorrhizae, ecto-mycorrhizae, and beneficial bacteria. When used, the species that are right for a given soil and climate will adapt and thrive. Soil inoculants increase the capacity of roots to absorb water and nutrients resulting in improved plant vigor, health, quality, and yield.
Whatever crop you are growing, enhancing biological activity in the soil vastly improves the crops capacity to reach its full genetic potential. Soils that are out of balance and lacking in needed minerals will most likely have a limited amount of biological activity. But when mineral nutrients are balanced and brought up to optimum levels, the fungi and bacteria will have something to work on and the soil will come alive. Using biological activators, or soil inoculants, will enhance and expand that activity by ensuring the bacteria and fungi that will best adapt to the soil and climate are present.
Bring your soil into balance with a custom soil prescription and amendment blend. Check out Step One to get started.
Nip It In The Bud And Nip It Before It Buds
Bromesedge, or Broomsedge as it is often called, is a member of the grass family. Its vegetative portion grows close to the ground producing light green, curly leaves. In the reproductive stage, Broomsedge produces a fibrous, golden stem that was often cut and bound together for use as a broom. However, its nutritive value as a forage is very low and grazing animals find it unpalatable.
We were discussing this problem with a local farmer a couple of weeks ago who runs a mixed herd of Scottish Highland and Irish Dexter beef cattle on his 34-acre farm near Odessa, Missouri.
Broomsedge is an opportunist. It capitalizes on situations of weakness in desirable forage crops. When a desirable plant is lost, it leaves a void in the canopy allowing more light to penetrate. This sets up the environment for Broomsedge to propagate and thrive.
The condition that most favors Broomsedge is poor soil fertility. Nutrient deficient pastures, especially with low levels of phosphorous and acidic soils, pH (<5.5) are a detriment to desirable forages and at the same time, provide the conditions for Broomsedge to invade. Phosphorous is very important to the maintenance of legumes in the pasture. So the first step in winning the battle with Broomsedge is to address nutrient deficiencies in the soil. This begins with a basic soil test.
Although the soil test will most likely show a Phosphorous deficiency, other mineral nutrients should not be ignored. Optimum soil fertility requires balancing the primary and secondary minerals. This is how we “nip it in the bud.” With the eleven primary and secondary minerals at optimum levels and ratios to each other, desirable plants will thrive and out-compete Broomsedge and remove the conditions favorable to its growth. If the budget is tight and you are unable to add all the minerals, begin with Calcium if it is required, and add the rest gradually over the next few years as recommended.
Things are not always as they seem. A Phosphorous deficiency may not mean a lack of Phosphorous. When the mineral nutrients are out of balance, the availability of Phosphorous may be affected even though there may be plenty of it in the soil. A shortage of one nutrient may “tie up” several others. This possibility emphasizes the importance of a soil test. You must know where you are if you are to get where you want to go.
We have nipped it in the bud by addressing soil fertility. So how do we “nip it before it buds?” Cattle may actively graze on the palatable, young Broomsedge vegetative growth occurring early in the season. Defoliation at this stage will reduce its vigor so early season grazing pressure can be part of the solution when Broomsedge is already a problem. On the other hand, inadequate defoliation pressure reduces stress on the Broomsedge and increases stress on desirable forage plants, giving Broomsedge the competitive advantage.
Other methods that have been used as a means of controlling and removing Broomsedge have their caveats. Burning can improve the regrowth quality of forage but Broomsedge is a warm season grass not damaged by early spring fires. Repeated burning on cool season pastures will eventually reduce the desired plants.
Any herbicide that would be effective against Broomsedge would also damage desirable plants. Targeted spot spraying may be possible but that would defeat the purpose of farming organically.
Mowing is really not effective because by the time Broomsedge is high enough to mow, it has reached the reproductive stage. Mowing would just facilitate seed disbursement.
Addressing soil fertility and evaluating grazing strategy are the most effective means of keeping Broomsedge out of the pasture and eliminating it when it becomes a problem. It took time for Broomsedge to gain a foothold and it will take time to get it under control. Optimizing soil fertility and patience will be rewarded with vigorous, nutrient-dense, Broomsedge-free forage.
Alas, the “Great Salt Myth.” Are salts created equal? Not quite. Commercially manufactured iodized salt is heavily processed and highly refined to remove impurities. Those impurities however are the trace minerals our body needs. Further degrading the quality, anti-caking agents have been added for uniform pouring since the early 1900s.
Table salt is 99% sodium chloride and 1% trace minerals. In contrast, Redmond Real Salt, mined underground from an ancient seabed that is now central Utah, is 97% sodium chloride and 3% trace minerals. That 3% contains 60 trace minerals. Himalayan sea salt, from an ancient seabed protected by lava, is 86% sodium chloride and 14% trace minerals of which there are 80+. What does all this mean? Reducing intake of processed foods high in sodium makes room for the use of mineral rich sea salts for most people. Our bodies really do require sodium for many important functions and benefits from the many trace minerals. There are exceptions, so it is imperative you check with your doctor.
Regarding cholesterol, the nutrient-dense animal foods listed in part three of the Mineral Message comprise 25% of the 1,100 to 1,700 milligrams of cholesterol we may have in our blood on a given day. The remaining 75% is actually produced inside our bodies by the liver. It has been shown that dietary cholesterol has very little impact on blood cholesterol levels in 75% of the population. The other 25% are referred to as hyper-responders with their cholesterol showing a moderate increase in LDL and HDL but does not affect the ratio of LDL to HDL or increase the risk of heart disease. Although other risk factors should not be dismissed, evidence suggests that the LDL particle number is a much better marker of heart disease risk than LDL level or total cholesterol level.
I originally planned to “examine more closely” the fact that if nutrient minerals are not in the soil, they can’t be in our bodies. It is such a simple fact but most of us are just not aware of the importance of our soils and how they serve as “the plants’ stomach.” By feeding the soil rather than the plant, we can ensure we get an abundance of the mineral nutrients we need. It all begins with a basic soil test indicating the levels and ratios of 11 mineral elements found in all soils. When we know where we are, we can determine where we need to go to balance these minerals for optimum nutrient density and flavor. Whether you are a home produce gardener, an organic farmer, or a rancher, healthy soil means healthy people and animals.
For further reading and research citations, see http://chriskresser.com/popular-posts/.
Chris Kresser, M.S., L.Ac is a globally recognized leader in the fields of ancestral health, Paleo nutrition, and functional and integrative medicine. Chris was recently named by Greatist.com as one of the 100 most influential people in health and fitness, along with Michelle Obama, Michael Pollan, Dr. Andrew Weil, Tim Ferriss, Mark Sisson, Robb Wolf and Dr. Mercola—and his work is frequently cited in national publications such as Time, The Atlantic, and NPR.
It has been said that we are what we eat. A more accurate statement might be we are what we absorb from what we eat. The truth is, animal fats help restore greater gut health, which increases the absorption rate of minerals and vitamins. That brings us to the fat-soluble vitamins A, D, E, and K.
Eggs yolks for example, are rich in vitamin A, beta-carotene, choline, Selenium, Manganese, as well as pantothenic acid, and vitamins, D and K. Choline is necessary for brain building and reproductive function. More Magnesium and Niacin are found in the egg whites. We need both the egg yolk and the egg whites for complete nutrition.
Let’s examine a few other foods on our ostensibly taboo list from part two. Vitamin E works as an antioxidant at the cellular level and is found in unprocessed oils as well as in animal fats like butter and eggs. True vitamin A is found only in animal foods such as cod liver oil, fish and shellfish, butter, eggs, and liver from pasture fed animals. Vitamins B6 and Bsub>12 are found only in animal foods. Vitamin D, required for mineral absorption, is found in organ meats, butterfat, and eggs from pasture fed animals. The diets of our ancestors contained ten times more vitamin D than the typical American diet today.
Minerals like Magnesium and Zinc are vital components of enzymes and are more easily absorbed from animal fats. CO-Enzyme Q1o, highly protective against cancer, is found only in animal foods. Cholesterol is a potent antioxidant that protects cell membranes against free radicals.
Bone broths are a source of much-needed minerals and collagen, and nourish the connective tissues throughout our bodies. Raw milk and cheese from pasture fed cows are rich sources of Calcium.
For those who are lactose intolerant, soured dairy products such as yogurt, sour cream, and ghee, (clarified butter from which the milk solids have been removed) are more easily digestible.
Finally, the traditional diet of our ancestors included meat and dairy from pastured animals. These animal products are nutritionally superior to products from grain fed ruminants that require antibiotics and other medications to relieve bloating, and that suffer other diseases resulting from unsanitary and cramped confinement.
In the fourth and final part of The Mineral Message, we will dispel the great salt myth, summarize how these aforementioned foods are not our enemies, and examine more closely the fact that if it’s not in the soil, it can’t be in the food.
In part one we looked at just one of the many health problems caused by malnutrition. If the minerals we require are not in the soil, they cannot be in our food nor can they be in our bodies. Without those mineral nutrients in adequate quantities, our bodies cannot synthesize the vitamins, amino acids, and proteins needed for a strong immune system. How do we get those nutrient minerals into our bodies? Quite easily!
We can begin by choosing whole foods grown and raised organically, but let’s not stop at just organic. We must go beyond organic to food produced in a soil that is in optimum mineral balance. It is then our bodies will have luxury amounts of the stuff strong immune systems are made of. Through modern research coupled with thousands of years of experience from our ancestors, the answers are easier and closer than you think.
Raw milk, cream, sour cream, cheese
Eggs from pasture raised chickens
Bacon and pork from pasture raised pigs
Grass fed and finished beef
Organ meats
Bone broth
Organic fruits and vegetables raised in minerally balanced soil
Cholesterol (Yes, cholesterol!)
Salt (Indeed, salt too!)
In part three we’ll examine the foods above, the nutrients and compounds they contain, and why they are important to our health.
To begin learning more about these foods and how they nurture us:
Nourishing Traditions: The Cookbook That Challenges Politically Correct Nutrition and the Diet Dictocrats by Sally Fallon, Mary Enig, and Marion Dearth. Or watch Dr. Kaayla Daniel on YouTube: https://www.youtube.com/user/DrKaaylaDaniel